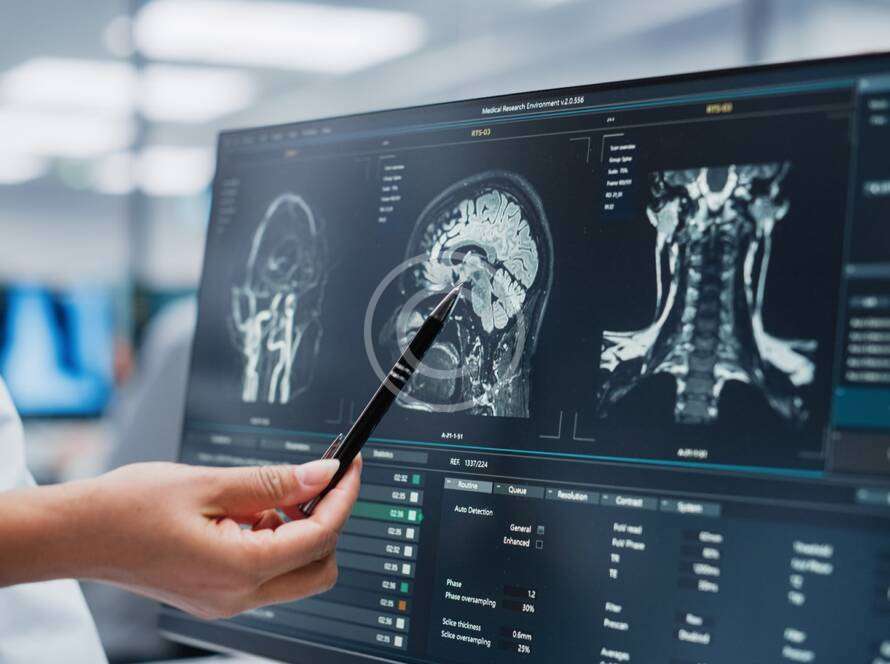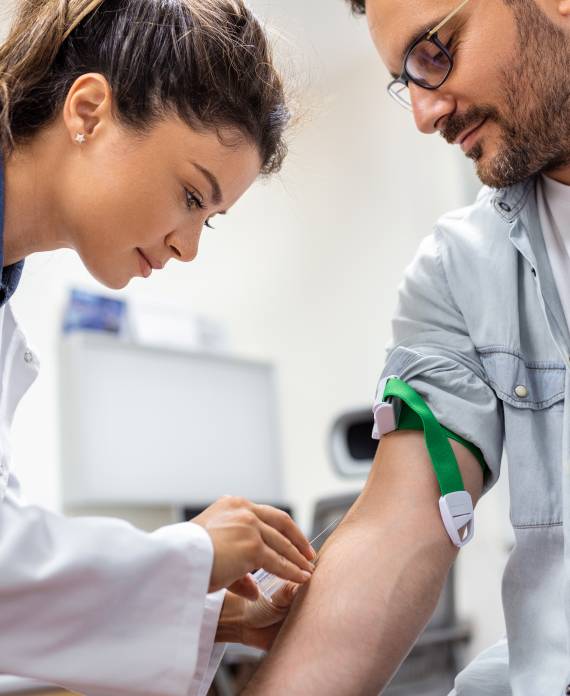
Why choose Folio?
Folio Clinical Research (Folio) is a Site Management Organization (SMO) based in southern California with 12 locations strategically scattered throughout the region. We offer operational, regulatory and other centralized research support services for sites on industry-sponsored trials.
We specialize in Phase I – IV urology and oncology trials in partnership with community physicians that treat thousands of patients each year. Our physician partners have participated as Principal and Sub-Investigators in a variety of studies for prostate cancer, bladder cancer, benign prostatic hyperplasia (BPH), urinary tract infections, and overactive bladder, among others.
Many private practice physicians have a desire to participate in research but aren’t sure how to get started. Folio makes it easier to get a clinical research program off the ground by providing staff, sourcing studies, and lending its expertise to establish a high-functioning site.
Folio has worked with many different sponsors, offering a highly efficient start-up process in stark contrast to academic institutions and hospitals. We offer sponsors access to a diverse patient population across our entire network.
Sites
Providers
Active Studies
States

CEO

COO

VP Clinical Operations

Director Contracts and Budgets

Senior Director, Business Development

VP Finance
Meet our leadership team
Partner Locations
Target identification
Compound screening
Preclinical testing
Have questions?
Get in touch!
Are you a practice owner interested in clinical research?
Are you an industry sponsor looking for additional sites?
Are you a patient looking to enroll in a clinical trial?
Please fill out the form to request more information or inquire about a potential collaboration.